History of Hot-Dip Galvanizing
- Share
- publisher
- Gloria Wang
- Issue Time
- Sep 1,2017
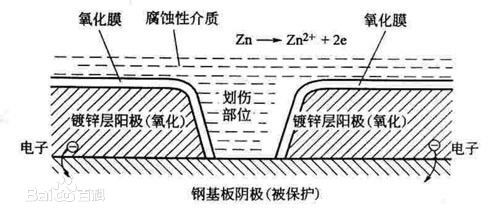
79 AD: Recordof zinc usage in construction began, which could be considered the originationof galvanizing.
1742: The first recorded history of galvanizing dates back to when P.J.Malouin, a French chemist, presented to the Royal Academy of Sciences severalexperiments involving coating iron with molten zinc.
1772:Luigi Galvani, galvanizing's namesake, discoveredthe electrochemical process that takes place between metals during anexperiment with frog legs.
1801: Alessandro Volta furthered the research on galvanizing when hediscovered the electro-potential between two metals, creating a corrosion cell.
1829:Michael Faraday discovered zinc's sacrificialaction during an experiment involving zinc, salt water, and nails.
1837: French engineer Stanislaus Tranquille Modeste Sorel took out apatent for the early galvanizing process.
1850: The British galvanizing industry was consuming 10,000 tons of zincannually for the production of galvanized steel.
1870: The United States, slightly behind, had its first galvanizing plantopen. At the time, the steel was handdipped in the zinc bath.
Today:More than 600,000 tons of zinc is consumed annuallyin North America to produce hot-dip galvanized steel – 200,000 tons for afterfabrication (batch) process and 400,000 for the continuous galvanizing process.Galvanizing is found in almost every major application and industry where ironor steel is used. The utilities, chemical process, pulp and paper, automotive,and transportation industries, to name just a few, historically have madeextensive use of galvanizing for corrosion control. They continue to do sotoday. Hot-dip galvanizing has a proven and growing history of success inmyriad applications worldwide.
